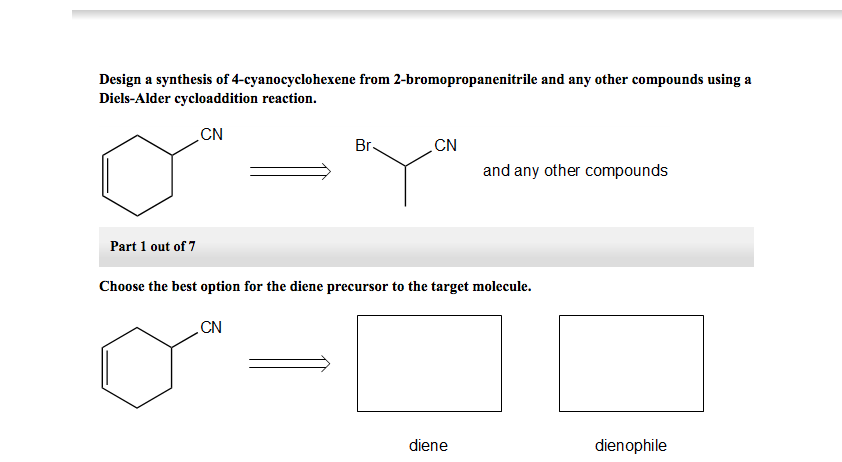
In this particular example, the epoxide shown undergoes rDA at 0 ☌. Release of nitrogen from six-membered, cyclic diazenes is common and often spontaneous at room temperature. The isocyanates may be isolated or trapped if they are the desired product. Release of isocyanates from Diels-Alder adducts of pyridones can be used to generate highly substituted aromatic compounds. The resulting highly substitued pyridines can be difficult to access by other means. Nitriles may be released in rDA reactions of DA adducts of pyrimidines or pyrazines. Highly reactive cyanoacrylates may be isolated from Diels-Alder adducts (synthesized independently) with the use of a scavenger. The scavenger reacts with either the diene or (more typically) the dienophile to drive the equilibrium of the retro DA process toward products. Alternatively, the rDA reaction may be carried out in the presence of a scavenger. Flash vacuum pyrolysis of Diels-Alder adducts synthesized by independent means can provide extremely reactive, short-lived dienophiles (which can then be captured by a unique diene). If isolation or reaction of an elusive diene or dienophile is the goal, one of two strategies may be used. Strategically placed electron-withdrawing groups in the starting material can render this process essentially irreversible. This strategy has been used, for instance, to generate aromatic cyclopentadienyl anions from adducts of cyclopentadiene. 
(3)īase mediation can be used to induce rDA in cases when the separated products are less basic than the starting material. Complexation of Lewis acids to basic functionality in the starting material may induce the retro Diels-Alder reaction, even in cases when the forward reaction is intramolecular. However, a variety of strategies for overcoming this inherent thermodynamic bias are known. The relative tendencies of a variety of dienes and dienophiles to form via rDA are described below:ĭiene: furan, pyrrole > benzene > naphthalene > fulvene > cyclopentadiene > anthracene > butadieneĭienophile: N 2 CO 2 > naphthalene > benzene, nitriles > methacrylate > maleimides > cyclopentadiene, imines, alkenes > alkynesīecause the Diels-Alder reaction exchanges two π bonds for two σ bonds, it is intrinsically thermodynamically favored in the forward direction. Most, however, require either thermal or chemical activation. Scope and LimitationsĪ few rDA reactions occur spontaneously at room temperature because of the high reactivity or volatility of the emitted dienophile. Much less is known about the relative rates of reversion of endo and exo adducts, and studies have pointed to no correlation between relative configuration in the cyclohexene starting material and reversion rate. 
Like the Diels-Alder reaction, the rDA preserves configuration in the diene and dienophile. Evidence for the latter was provided by the reaction below-none of the "head-to-head" isomer was obtained, suggesting a fully intramolecular isomerization process. However, such isomerization may take place via a completely intramolecular, -sigmatropic (Cope) process. It was postulated that at high temperatures, isomerization of kinetic endo adducts to more thermodynamically stable exo products occured via an rDA/DA sequence. Evidence for the retro-Diels-Alder reaction was provided by the observation of endo- exo isomerization of Diels-Alder adducts. Can you explain this answer? tests, examples and also practice Chemistry tests.The retro Diels-Alder reaction proper is essentially the microscopic reverse of the Diels-Alder reaction: a concerted (but not necessarily synchronous), pericyclic, single-step process. Can you explain this answer? theory, EduRev gives you anĪmple number of questions to practice Order of reactivity of the following dienes X, Y and Z in Diels-Alder reaction is: a)X > Z > Yb)Y > X > Zc)Y > Z > Xd)X > Y > ZCorrect answer is option 'D'. Can you explain this answer? has been provided alongside types of Order of reactivity of the following dienes X, Y and Z in Diels-Alder reaction is: a)X > Z > Yb)Y > X > Zc)Y > Z > Xd)X > Y > ZCorrect answer is option 'D'. Can you explain this answer?, a detailed solution for Order of reactivity of the following dienes X, Y and Z in Diels-Alder reaction is: a)X > Z > Yb)Y > X > Zc)Y > Z > Xd)X > Y > ZCorrect answer is option 'D'. Order of reactivity of the following dienes X, Y and Z in Diels-Alder reaction is: a)X > Z > Yb)Y > X > Zc)Y > Z > Xd)X > Y > ZCorrect answer is option 'D'. 
Can you explain this answer? defined & explained in the simplest way possible. Here you can find the meaning of Order of reactivity of the following dienes X, Y and Z in Diels-Alder reaction is: a)X > Z > Yb)Y > X > Zc)Y > Z > Xd)X > Y > ZCorrect answer is option 'D'.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
